[ad_1]
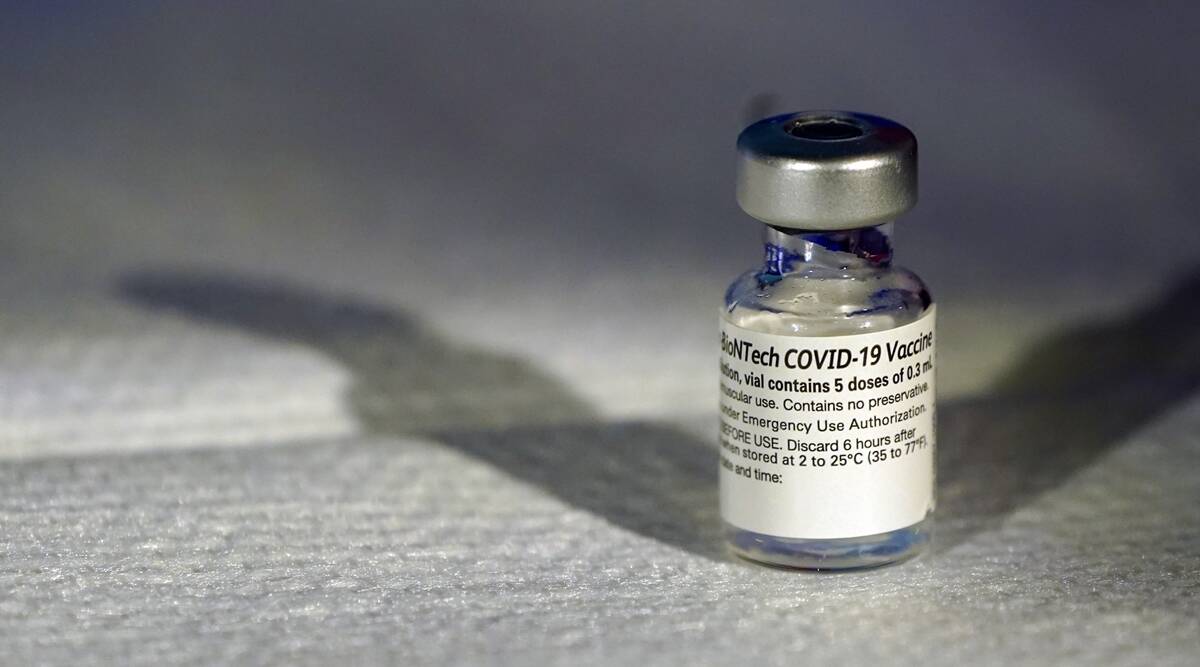
 A vial of Pfizer’s COVID-19 vaccine that receivedemergency use authorization is seen at George Washington University Hospital, Monday, Dec. 14, 2020, in Washington. (AP)
A vial of Pfizer’s COVID-19 vaccine that receivedemergency use authorization is seen at George Washington University Hospital, Monday, Dec. 14, 2020, in Washington. (AP)
As the Covid-19 pandemic continues to wreak havoc globally, with one other wave anticipated in Europe throughout Christmas, a minimum of 8 countries have begun or granted approval for vaccinating their folks in opposition to the virus.
On November 18, the vaccine developed by US producer Pfizer and German biotechnology firm BioNTech grew to become the primary on the earth to launch full late-stage trial information. Britain was the primary to approve the shot for emergency use on December 3, adopted by Canada on December 9 and the US Food and Drug Administration (FDA) on December 11. Apart from Pfizer, Moderna grew to become a detailed second in lots of countries after it launched a full information evaluation for a late-stage trial on November 30 exhibiting a 94.1 per cent efficacy fee for its vaccine.
As folks internationally start to roll up their sleeves for the coronavirus vaccine, here’s a have a look at which nation has authorised which vaccine
Switzerland
Following a two-month-long overview, Switzerland Saturday grew to become the most recent nation to approve the Pfizer-BioNTech vaccine to be used.
“After a meticulous review of the available information, Swissmedic concluded that the Covid-19 vaccine from Pfizer/BioNTech is safe and that its benefit outweighs the risks,” the Swissmedic regulatory authority stated in an announcement.
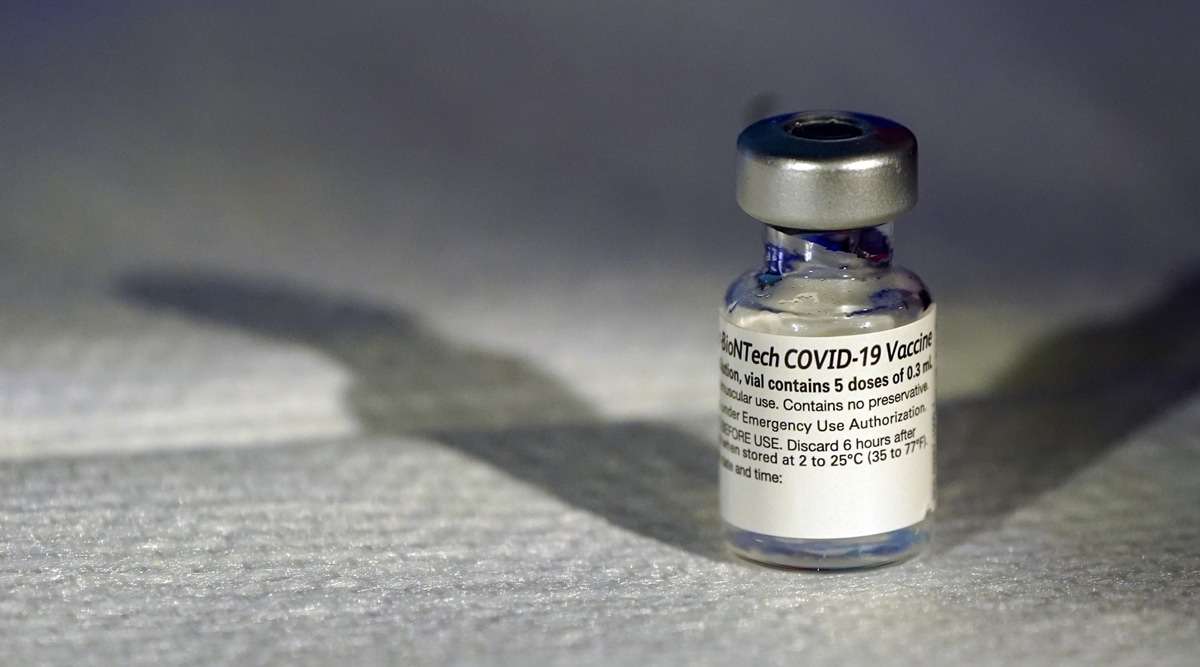
 A vial of the Pfizer-BioNTech COVID-19 vaccine. (AP)
A vial of the Pfizer-BioNTech COVID-19 vaccine. (AP)
It is the primary vaccine in opposition to the brand new coronavirus authorised to this point within the nation.
According to AFP, the alpine nation has secured round 15.Eight million Covid-19 vaccine doses, in offers with three separate producers. It has signed contracts for round three million doses of the Pfizer-BioNTech vaccine, round 7.5 million doses of Moderna’s vaccine, and round 5.Three million doses of the AstraZeneca vaccine. All three vaccines require two doses per individual.
United States
The United States Friday added a second Covid-19 vaccine to its arsenal after FDA authorised an emergency rollout of the vaccine developed by Moderna Inc. and the National Institutes of Health.
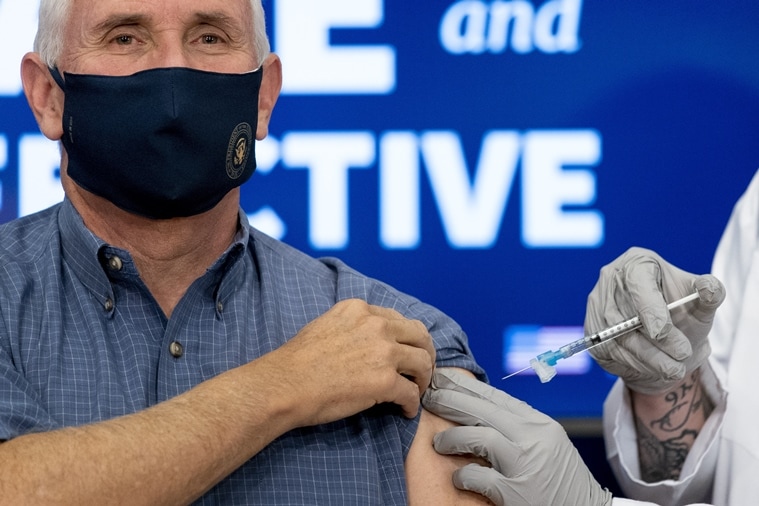 Vice President Mike Pence receives a Pfizer-BioNTech COVID-19 vaccine shot on the Eisenhower Executive Office Building on the White House advanced, Friday, Dec. 18, 2020, in Washington. (AP)
Vice President Mike Pence receives a Pfizer-BioNTech COVID-19 vaccine shot on the Eisenhower Executive Office Building on the White House advanced, Friday, Dec. 18, 2020, in Washington. (AP)
The transfer additionally marks the world’s first authorisation for Moderna’s Covid-19 vaccine. According to the Associated Press, the vaccine is similar to one from Pfizer Inc. and Germany’s BioNTech that’s now being disbursed to thousands and thousands of healthcare employees and nursing house residents as the most important vaccination drive in US historical past begins to ramp up.
Compared to Pfizer, the vaccine produced by Moderna’s is simpler to deal with because it doesn’t should be saved at ultra-freezing temperatures. The vaccine might be saved for as much as six months at -20 levels Celsius, although it’s anticipated to be steady for 30 days at regular fridge temperature of two to eight levels Celsius (36 levels Fahrenheit to 46 levels Fahrenheit). Early outcomes of enormous, nonetheless unfinished research present each vaccines seem secure and strongly protecting.
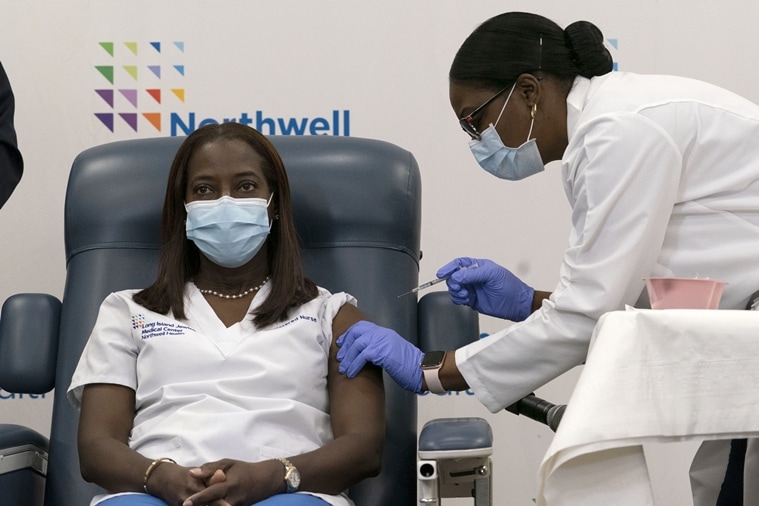 Sandra Lindsay, left, a nurse at Long Island Jewish Medical Center, is inoculated with the Pfizer-BioNTech COVID-19 vaccine by Dr. Michelle Chester, Monday, Dec. 14, 2020, within the Queens borough of New York. (AP)
Sandra Lindsay, left, a nurse at Long Island Jewish Medical Center, is inoculated with the Pfizer-BioNTech COVID-19 vaccine by Dr. Michelle Chester, Monday, Dec. 14, 2020, within the Queens borough of New York. (AP)
The United States to this point has been the worst-affected nation by the coronavirus pandemic. The scourge has claimed greater than 3,12,000 US lives and 1.7 million folks worldwide. New instances within the US are working at over 216,000 per day on common. Deaths per day have hit all-time highs, eclipsing 3,600 on Wednesday.
Malaysia
Malaysia on Saturday stated it expects to obtain the primary batch of the Covid-19 vaccine collectively developed by Pfizer and BioNTech in February, information company Reuters reported.
 A clinic physician collects pattern for coronavirus testing from motorcyclist at a COVID-19 screening facility at Clinic Ajwa close to a mural depicting medical frontlines in Shah Alam, Selangor state, Malaysia, on Saturday, Dec. 12, 2020. (AP)
A clinic physician collects pattern for coronavirus testing from motorcyclist at a COVID-19 screening facility at Clinic Ajwa close to a mural depicting medical frontlines in Shah Alam, Selangor state, Malaysia, on Saturday, Dec. 12, 2020. (AP)
In November, Malaysia had introduced that it had agreed to purchase 12.Eight million doses of the vaccine, turning into the primary nation in Southeast Asia to strike a cope with the US drugmaker. Under the deal, Pfizer will ship the primary a million doses within the first quarter of 2021, with 1.7 million, 5.Eight million and 4.Three million doses to comply with in subsequent quarters.
The Malaysian authorities additionally stated that they’re in talks with different pharmaceutical corporations to safe extra vaccines.
“The government is making efforts to get a bigger supply of vaccine to meet our needs. The government will take the advice of the Health Ministry regarding the percentage of people who need to be vaccinated,” Trade Minister Mohamed Azmin was quoted as saying by Reuters.
Britain
On December 8, the United Kingdom grew to become the primary nation on the earth to manage Covid-19 vaccine to its public. The first recipient of the vaccine developed by Pfizer-BionTech was 90-year-old Margaret Keenan. She obtained the shot at University Hospital Coventry at 6:31 a.m. Keenan stated she felt “so privileged to be the primary individual vaccinated in opposition to Covid-19.
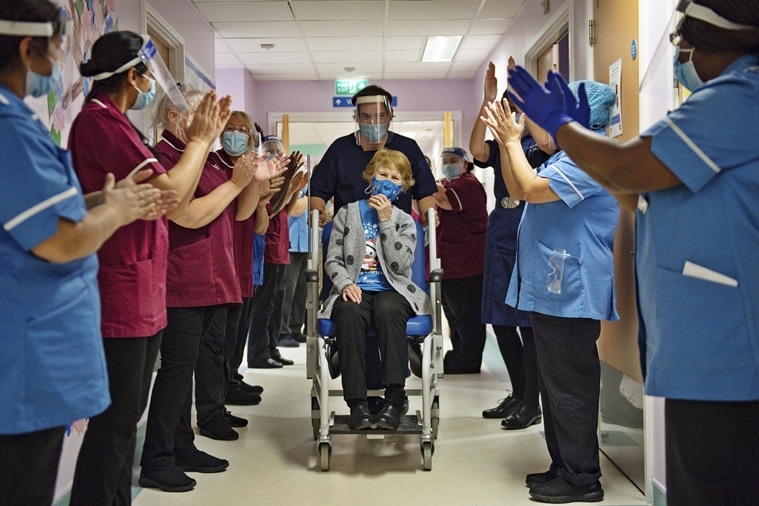 Margaret Keenan, 90, is applauded by employees as she returns to her ward after turning into the primary affected person within the UK to obtain the Pfizer-BioNTech COVID-19 vaccine, at University Hospital, Coventry, England, Tuesday Dec. 8, 2020. (AP)
Margaret Keenan, 90, is applauded by employees as she returns to her ward after turning into the primary affected person within the UK to obtain the Pfizer-BioNTech COVID-19 vaccine, at University Hospital, Coventry, England, Tuesday Dec. 8, 2020. (AP)
According to BBC, it was the primary of 800,000 doses of the vaccine that will probably be disbursed within the coming weeks.
In England, the vaccine will probably be delivered at 50 hospital hubs within the first wave of the programme, with extra hospitals anticipated to supply it because the rollout ramps up. Northern Ireland, Scotland and Wales are making their very own plans underneath the UK’s system of the devolved administration.
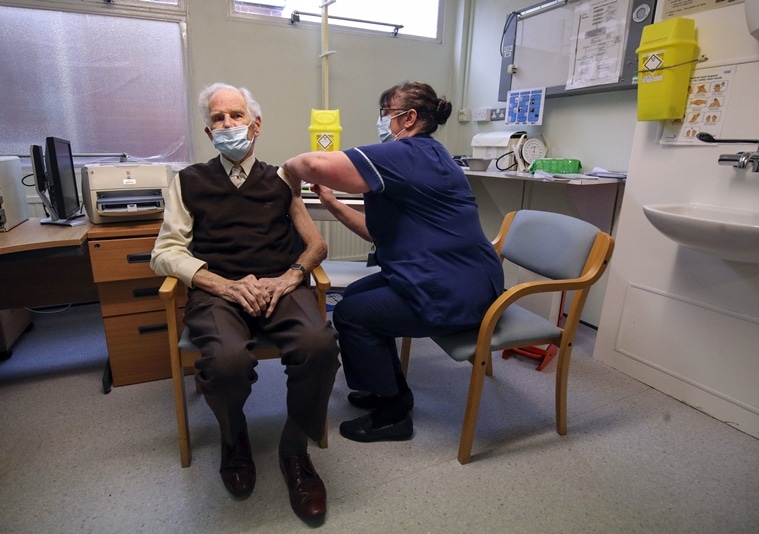 Brian Horne receives the Pfizer BioNtech COVID-19 vaccine in Chalfont St Peter, England, Monday Dec. 14, 2020. (AP)
Brian Horne receives the Pfizer BioNtech COVID-19 vaccine in Chalfont St Peter, England, Monday Dec. 14, 2020. (AP)
The nation has positioned orders for practically 40 million doses — sufficient for 20 million folks, as two programs are wanted. However, most provides are usually not anticipated to turn into accessible till subsequent yr.
Another vaccine being developed by Oxford-AstraZeneca within the UK has additionally been discovered to be “safe and effective” and is being at the moment assessed by unbiased scientists.
Bahrain
Following in Britain’s footsteps, the island kingdom of Bahrain on December Four grew to become the second nation on the earth to grant an emergency-use authorisation for the coronavirus vaccine made by Pfizer-BioNTech. The nation has provided residents the choice to pre-register to obtain the vaccine on its web site.
 A droplet falls from a syringe after a well being care employee is injected with the Pfizer-BioNTech COVID-19 vaccine. (AP)
A droplet falls from a syringe after a well being care employee is injected with the Pfizer-BioNTech COVID-19 vaccine. (AP)
On December 13, the nation additionally introduced the approval of a vaccine developed by Sinopharm Group, a Chinese government-owned pharmaceutical firm. According to a report in The Guardian, Sinopharm is amongst two Chinese pharmaceutical corporations (the opposite is fellow frontrunner Sinovac) to have created their vaccine by way of the extra conventional technique of utilizing an inactive virus to set off an immune response.
Sinopharm’s shot depends on a examined know-how, utilizing a killed virus to ship the vaccine, much like how polio immunizations are made. Leading Western opponents, just like the shot made by Pfizer and its German associate BioNTech, use newer, less-proven know-how to focus on the coronavirus’ spike protein utilizing RNA.
 File: Pfizer vaccine picture illustration
File: Pfizer vaccine picture illustration
However, the nation provided few particulars on examine outcomes of the vaccine, according to the United Arab Emirates, which final week introduced the vaccine was 86% efficient. According to the Bahrain authorities, over 7,700 folks signed up to participate in a trial of the Sinopharm vaccine within the kingdom.
Canada
In an effort to beat the pandemic, Canada administered its first doses of a Covid-19 vaccine on December 14. Five front-line employees in Ontario have been among the many first Canadians to obtain the Pfizer-BioNtech vaccine at considered one of Toronto’s hospitals.
The Canadian authorities had just lately amended its contract with Pfizer and BioNTech in order that it will ship as much as 249,000 doses in December itself. As per AP, extra of Canada’s preliminary 30,000 doses of the vaccine are anticipated to cross the border by Monday.
Anita Quidangen, a private help employee who labored all through a COVID-19 outbreak on the Rekai Centre nursing house in Toronto, bought the primary dose in Ontario.
 Canada Prime Minister Justin Trudeau and a person who had simply obtained the Pfizer-BioNTech COVID-19 vaccine elbow bump because the Prime Minister toured a vaccinations clinic on the Ottawa Hospital with Minister of Health Patty Hajdu, Tuesday, Dec. 15, 2020, in Ottawa. (AP)
Canada Prime Minister Justin Trudeau and a person who had simply obtained the Pfizer-BioNTech COVID-19 vaccine elbow bump because the Prime Minister toured a vaccinations clinic on the Ottawa Hospital with Minister of Health Patty Hajdu, Tuesday, Dec. 15, 2020, in Ottawa. (AP)
The nation has contracts with six different vaccine makers and is at the moment reviewing three different vaccines, together with one by Moderna that Canadian well being officers stated could possibly be authorised quickly.
Canada has ordered extra doses than wanted for Canadians however the authorities finally plans to donate extra provide to impoverished countries.
Mexico
Following Britain, Canada and Bahrain, Mexico grew to become the fourth nation to approve emergency use of Pfizer’s COVID-19 vaccine, following authorisation from well being regulator Cofepris on Friday.
The Mexican authorities has inked an settlement with Pfizer to amass 34.Four million doses of its vaccine, with the primary batch anticipated to reach this month.
The nation has to this point reported 1.Three million coronavirus an infection and over 1,17,000 deaths.
Russia and China
While Pfizer’s shot was the primary to be rolled out following the publication of full Phase III trial information, Russia and China have been inoculating their residents for months with a number of completely different vaccines nonetheless present process late-stage trials.
On November 24, Russia stated that its Sputnik V vaccine, developed by the Gamaleya Institute, was 91.Four per cent efficient based mostly on interim late-stage trial outcomes. It started vaccinations in August and has inoculated greater than 100,000 folks to this point.
 People sporting face masks to assist curb the unfold of the coronavirus stroll by an ethical depicting China’s skyscrapers alongside a road in Beijing, Sunday, Dec. 6, 2020. (AP)
People sporting face masks to assist curb the unfold of the coronavirus stroll by an ethical depicting China’s skyscrapers alongside a road in Beijing, Sunday, Dec. 6, 2020. (AP)
On the opposite hand, China on December 18 introduced its plan to vaccinate 50 million folks within the high-priority group in opposition to the coronavirus earlier than the beginning of the height Lunar New Year journey season early subsequent yr.
The nation has to this point granted emergency-use standing to 2 candidate vaccines from Sinopharm and one from Sinovac Biotech. It has additionally authorised a fourth, from CanSino Biologics Inc, for navy use.
According to a report printed within the South China Morning Post, Chinese officers have been requested to finish the primary 50 million doses by January 15 and the second by February 5.
(Inputs from businesses)
[ad_2]
Source hyperlink