[ad_1]
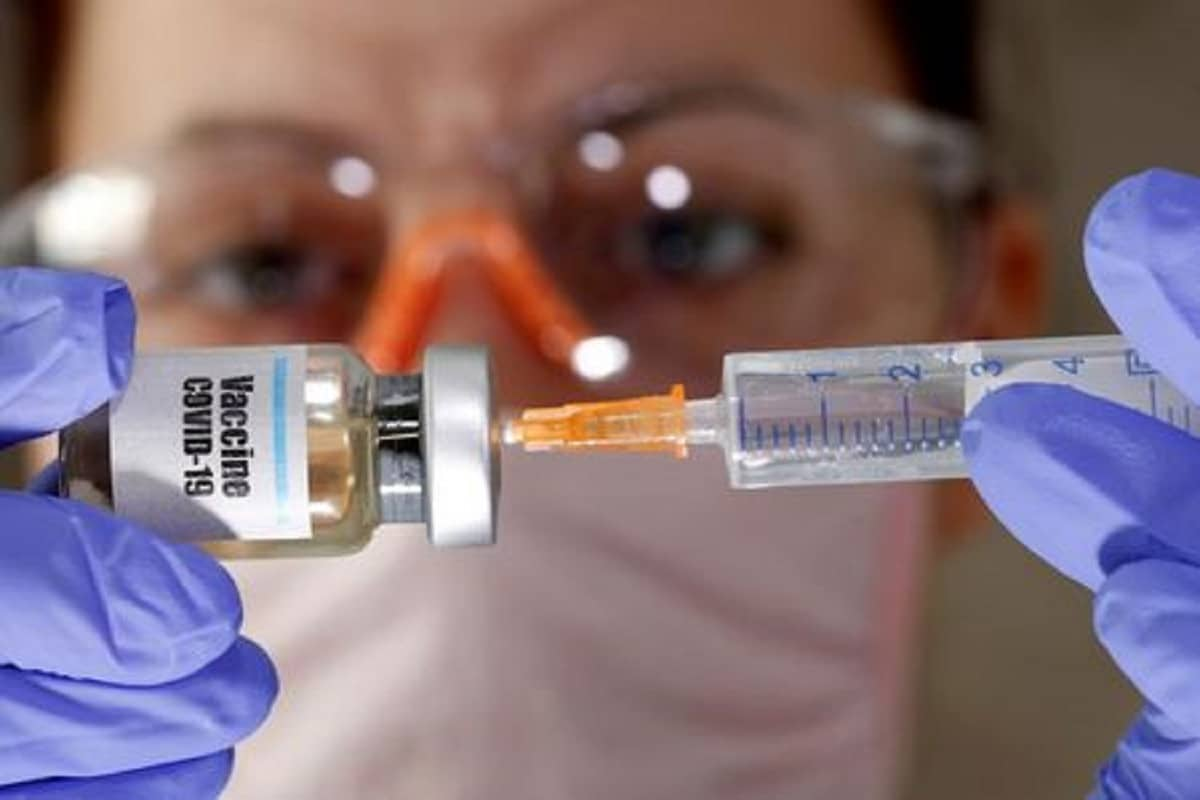
The third part human trial of Covaxin, the much-awaited indigenously developed Covid-19 vaccine, has begun at an institute right here, a senior official stated on Friday. The vaccine was administered to 2 recruits on Thursday on the Preventive and Therapeutic Clinical Trial Unit (PTCTU) on the Institute of Medical Sciences and SUM Hospital, the one institute in Odisha chosen by the Indian Council for Medical Research (ICMR) for the human trial of the vaccine, E.Venkata Rao, the principal investigator in Covaxin human trial stated.
vaccine, has begun at an institute right here, a senior official stated on Friday. The vaccine was administered to 2 recruits on Thursday on the Preventive and Therapeutic Clinical Trial Unit (PTCTU) on the Institute of Medical Sciences and SUM Hospital, the one institute in Odisha chosen by the Indian Council for Medical Research (ICMR) for the human trial of the vaccine, E.Venkata Rao, the principal investigator in Covaxin human trial stated.
The indigenous vaccine, which is being developed by Bharat Biotech and ICMR, has obtained approval of the Central Drugs Standard Control Organisation (CDSCO) for initiating the third part trial.The trial is being carried out at 21 chosen medical institutes throughout the nation.
Rao stated the vaccines first part trial was aimed toward measuring its security, whereas part two was meant to check its immunogenicity. The third part trial will examine the efficacy of the vaccine, he stated including security examine (Phase 1) primarily appeared on the security side for human use with none vital facet impact.
Immunogenicity examine (Phase 2) measured the antibody degree within the human blood and checked whether or not it was adequate to guard the individual from the an infection, he stated. Phase three will consider if the vaccine was truly capable of forestall improvement of the illness among the many recipients of the vaccine, he stated.
In truth part three of the trial is essential and tedious as we’ve got to attend until the event of the illness which depends on the frequency of the illness within the inhabitants. So we have to recruit a big quantity of topics and greater than 25,000 volunteers have been being recruited for the trial throughout the nation, he stated. In part three two doses will likely be administered to every volunteer 28 days aside. The observe up interval will lengthen to 12 months to have a look at its long run and negative effects, Rao, who’s a professor within the division of group drugs at IMS and SUM Hospital, stated.
Covaxin, he stated, is an inactivated complete cell virion with out the flexibility to provide the illness however helps in growing antibodies towards the an infection. A virion is a whole virus particle consisting of an outer protein shell and an inside core of nucleic acid. The core confers ingectivity and the outer shell offers specificity to the virus.
These are time-tested vaccines whereas the others being developed have been mRNA vaccines, new to human inhabitants, which ship a sign into the human cell for the manufacturing of antibodies towards the illness. While recruiting volunteers for the part three trial, the restriction of being a wholesome volunteer was not obligatory. Anyone with diabetes or hypertension or with every other illness may additionally take part within the trial supplied the illness was underneath management in them with treatment, he stated.
Volunteers desirous of collaborating within the trial can register on-line or ship a message by WhatsApp with their identify, age, gender and the place of residence to 7849021450, he added.
.
[ad_2]
Source hyperlink