[ad_1]
 An indication that includes the AstraZeneca Plc emblem stands close to the corporate’s DaVinci constructing on the Melbourn Science Park in Cambridge, U.Ok., on Monday, June 8, 2020. (Photographer: Jason Alden/Bloomberg)
An indication that includes the AstraZeneca Plc emblem stands close to the corporate’s DaVinci constructing on the Melbourn Science Park in Cambridge, U.Ok., on Monday, June 8, 2020. (Photographer: Jason Alden/Bloomberg)
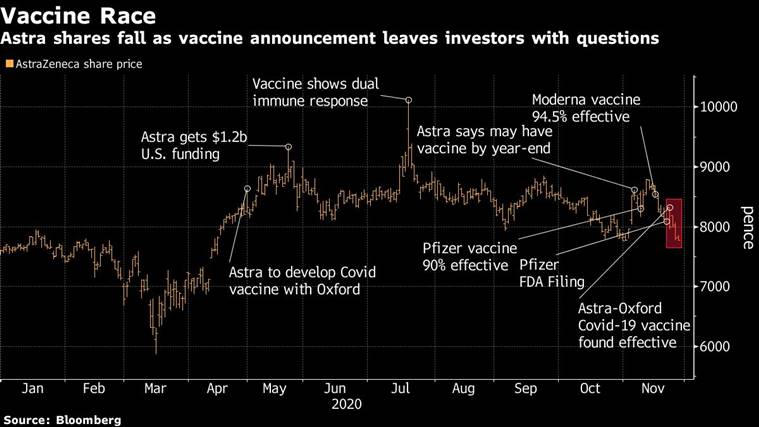
AstraZeneca Plc’s Covid-19 vaccine appears prefer it’s headed for a further global trial as the drugmaker tries to clear up uncertainty and confusion surrounding favorable ends in its present examine.
The firm needs the brand new take a look at to verify the 90% efficacy charge that the shot confirmed in a portion of an current trial, Chief Executive Officer Pascal Soriot mentioned. It’s favoring that choice relatively than including an arm to a separate examine that’s already underway within the U.S.
Questions are mounting over one of many fastest-moving photographs after the corporate acknowledged {that a} decrease dosage degree that appeared simpler resulted from a producing discrepancy. The firm and its companion, the University of Oxford, didn’t initially disclose the error and different key particulars, resulting in concern over their transparency.
“Now that we’ve found what looks like a better efficacy we have to validate this, so we need to do an additional study,” Soriot mentioned in his first interview because the knowledge have been launched. It will most likely be one other “international study, but this one could be faster because we know the efficacy is high so we need a smaller number of patients.”
Soriot mentioned he didn’t anticipate the extra trial to carry up regulatory approvals within the U.Ok. and European Union.
The U.Ok. authorities mentioned Friday it had requested the medicines regulator to evaluate whether or not the Astra-Oxford vaccine was appropriate for non permanent authorization. The uncommon step comes after the federal government amended laws in mild of the pandemic to permit the U.Ok. to approve a vaccine forward of the European regulator, which Britain remains to be topic to till January.
Clearance from the U.S. Food and Drug Administration might take longer as a result of the regulator is unlikely to approve the vaccine on the idea of research carried out elsewhere, particularly given the questions over the outcomes, Soriot mentioned. Authorization in some nations remains to be anticipated earlier than the tip of the yr, he mentioned.
“The question for us was, will we need the U.S. data to get approval in the U.S. or can we get approval in the U.S. with international data, and it was never clear,” mentioned Soriot, who’s in quarantine after arriving in Australia. “Now with those results it’s more likely that we will need the U.S. data.”
Astra and its CEO are going through scrutiny as the drugmaker responds to rising confusion over the vaccine. The firm’s late-stage knowledge initially elevated confidence that the world would quickly have a number of photographs to fight the pathogen, following constructive studies from front-runners Pfizer Inc. and Moderna Inc. But scant disclosures and the manufacturing discrepancy have sparked doubts amongst scientists and traders.
Different Rates
Astra and Oxford reported Monday {that a} decrease preliminary dose of the vaccine, adopted by a full dose, produced a 90% efficacy charge in a smaller set of members, in contrast with 62% for 2 full doses.
A day after the information have been unveiled the pinnacle of Operation Warp Speed, the U.S. vaccine program, mentioned that the routine exhibiting the upper degree of effectiveness was examined in a youthful inhabitants. He additionally mentioned the half-dose was given to some folks due to an error within the amount of vaccine put into some vials. None of these particulars have been disclosed in Astra or Oxford’s authentic statements.
Astra May Need New Covid-19 Vaccine Trial Beyond the U.S.: React
Soriot disputed the concept the half-dose routine was an error, saying that after researchers realized the dosing discrepancy they formally modified the trial protocol with the blessing of regulators.
“I won’t tell you we expected the efficacy to be higher,” mentioned Soriot. But “people call it a mistake — it’s not a mistake.”
Astra shares closed 0.7% decrease in London. They have declined about 7% this week amid questions concerning the trial outcomes.
The firm has beforehand mentioned it would add a brand new arm to its U.S. trial to check the decrease dosage.
Astra and researchers have declined to offer extra knowledge forward of a peer-reviewed evaluation that’s anticipated to be revealed within the coming weeks. Results have been submitted to an undisclosed journal, Astra mentioned in a press release.
Astra’s is considered one of three vaccines that might be accredited earlier than the tip of the yr. Pfizer and Moderna, which have each created photographs utilizing messenger RNA, revealed knowledge earlier this month that confirmed their vaccines have been about 95% efficient, and Pfizer has utilized to U.S. regulators for emergency approval.
There’s added strain on the Astra shot to succeed as a result of it’s simpler to retailer and the corporate is promoting it at value throughout the pandemic, which suggests many low- and middle-income nations are counting on it.
[ad_2]
Source hyperlink

